

You can:
| Name | Lysophosphatidic acid receptor 3 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | LPAR3 |
| Synonym | Lysophosphatidic acid receptor Edg-7 LPA3 receptor LPA-3 LPA receptor 3 endothelial differentiation gene 7, lysophosphatidic acid G-protein-coupled receptor 7 [ Show all ] |
| Disease | Fibrosis |
| Length | 353 |
| Amino acid sequence | MNECHYDKHMDFFYNRSNTDTVDDWTGTKLVIVLCVGTFFCLFIFFSNSLVIAAVIKNRKFHFPFYYLLANLAAADFFAGIAYVFLMFNTGPVSKTLTVNRWFLRQGLLDSSLTASLTNLLVIAVERHMSIMRMRVHSNLTKKRVTLLILLVWAIAIFMGAVPTLGWNCLCNISACSSLAPIYSRSYLVFWTVSNLMAFLIMVVVYLRIYVYVKRKTNVLSPHTSGSISRRRTPMKLMKTVMTVLGAFVVCWTPGLVVLLLDGLNCRQCGVQHVKRWFLLLALLNSVVNPIIYSYKDEDMYGTMKKMICCFSQENPERRPSRIPSTVLSRSDTGSQYIEDSISQGAVCNKSTS |
| UniProt | Q9UBY5 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | Q9UBY5 |
| 3D structure model | This predicted structure model is from GPCR-EXP Q9UBY5. |
| BioLiP | N/A |
| Therapeutic Target Database | T95923 |
| ChEMBL | CHEMBL3250 |
| IUPHAR | 274 |
| DrugBank | N/A |
| Name | CHEMBL2335050 |
|---|---|
| Molecular formula | C21H44NaO5PS |
| IUPAC name | sodium;(2-heptadecoxy-3-methoxypropoxy)-hydroxy-oxido-sulfanylidene-lambda5-phosphane |
| Molecular weight | 462.602 |
| Hydrogen bond acceptor | 6 |
| Hydrogen bond donor | 1 |
| XlogP | None |
| Synonyms | N/A |
| Inchi Key | JZDLPGVYJPWQNT-UHFFFAOYSA-M |
| Inchi ID | InChI=1S/C21H45O5PS.Na/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-25-21(19-24-2)20-26-27(22,23)28;/h21H,3-20H2,1-2H3,(H2,22,23,28);/q;+1/p-1 |
| PubChem CID | 71718248 |
| ChEMBL | CHEMBL2335050 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | N/A |
Structure | 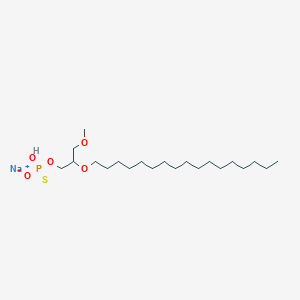 |
| Lipinski's druglikeness | Partition coefficient log P of this ligand is not available. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| EC50 | 0.39 nM | PMID23395664 | ChEMBL |
| EC50 | 0.3981 nM | PMID23395664 | ChEMBL |
| Intrinsic activity | 340.0 - | PMID23395664 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218