

You can:
| Name | Relaxin receptor 1 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | RXFP1 |
| Synonym | Relaxin family peptide receptor 1 RX1 relaxin/insulin like family peptide receptor 1 relaxin receptor 1 RXFPR1 [ Show all ] |
| Disease | Acute heart failure |
| Length | 757 |
| Amino acid sequence | MTSGSVFFYILIFGKYFSHGGGQDVKCSLGYFPCGNITKCLPQLLHCNGVDDCGNQADEDNCGDNNGWSLQFDKYFASYYKMTSQYPFEAETPECLVGSVPVQCLCQGLELDCDETNLRAVPSVSSNVTAMSLQWNLIRKLPPDCFKNYHDLQKLYLQNNKITSISIYAFRGLNSLTKLYLSHNRITFLKPGVFEDLHRLEWLIIEDNHLSRISPPTFYGLNSLILLVLMNNVLTRLPDKPLCQHMPRLHWLDLEGNHIHNLRNLTFISCSNLTVLVMRKNKINHLNENTFAPLQKLDELDLGSNKIENLPPLIFKDLKELSQLNLSYNPIQKIQANQFDYLVKLKSLSLEGIEISNIQQRMFRPLMNLSHIYFKKFQYCGYAPHVRSCKPNTDGISSLENLLASIIQRVFVWVVSAVTCFGNIFVICMRPYIRSENKLYAMSIISLCCADCLMGIYLFVIGGFDLKFRGEYNKHAQLWMESTHCQLVGSLAILSTEVSVLLLTFLTLEKYICIVYPFRCVRPGKCRTITVLILIWITGFIVAFIPLSNKEFFKNYYGTNGVCFPLHSEDTESIGAQIYSVAIFLGINLAAFIIIVFSYGSMFYSVHQSAITATEIRNQVKKEMILAKRFFFIVFTDALCWIPIFVVKFLSLLQVEIPGTITSWVVIFILPINSALNPILYTLTTRPFKEMIHRFWYNYRQRKSMDSKGQKTYAPSFIWVEMWPLQEMPPELMKPDLFTYPCEMSLISQSTRLNSYS |
| UniProt | Q9HBX9 |
| Protein Data Bank | 2jm4 |
| GPCR-HGmod model | N/A |
| 3D structure model | This structure is from PDB ID 2jm4. |
| BioLiP | BL0101296 |
| Therapeutic Target Database | T73696 |
| ChEMBL | CHEMBL1293316 |
| IUPHAR | 351 |
| DrugBank | N/A |
| Name | CHEMBL1562390 |
|---|---|
| Molecular formula | C17H13ClFN5 |
| IUPAC name | N-(3-chloro-4-fluorophenyl)-3-ethyl-[1,2,4]triazolo[4,3-c]quinazolin-5-amine |
| Molecular weight | 341.774 |
| Hydrogen bond acceptor | 5 |
| Hydrogen bond donor | 1 |
| XlogP | 5.3 |
| Synonyms | N-(3-chloro-4-fluorophenyl)-3-ethyl[1,2,4]triazolo[4,3-c]quinazolin-5-amine MCULE-6782618209 AKOS001804813 NCGC00107326-01 MolPort-007-615-961 [ Show all ] |
| Inchi Key | ACMFQRATSNZCDM-UHFFFAOYSA-N |
| Inchi ID | InChI=1S/C17H13ClFN5/c1-2-15-22-23-16-11-5-3-4-6-14(11)21-17(24(15)16)20-10-7-8-13(19)12(18)9-10/h3-9H,2H2,1H3,(H,20,21) |
| PubChem CID | 15990056 |
| ChEMBL | CHEMBL1562390 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | N/A |
Structure | 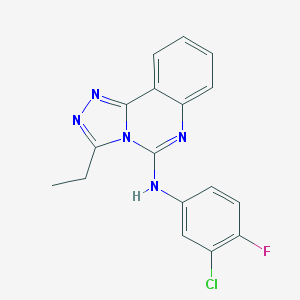 |
| Lipinski's druglikeness | This ligand has a partition coefficient log P greater than 5. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Potency | 39810.7 nM | PubChem BioAssay data set | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218