

You can:
| Name | Free fatty acid receptor 1 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | FFAR1 |
| Synonym | FFA1R G protein-coupled receptor 40 G-protein coupled receptor 40 GPR40 FFA1 receptor |
| Disease | Type 2 diabetes Non-insulin dependent diabetes Diabetes |
| Length | 300 |
| Amino acid sequence | MDLPPQLSFGLYVAAFALGFPLNVLAIRGATAHARLRLTPSLVYALNLGCSDLLLTVSLPLKAVEALASGAWPLPASLCPVFAVAHFFPLYAGGGFLAALSAGRYLGAAFPLGYQAFRRPCYSWGVCAAIWALVLCHLGLVFGLEAPGGWLDHSNTSLGINTPVNGSPVCLEAWDPASAGPARFSLSLLLFFLPLAITAFCYVGCLRALARSGLTHRRKLRAAWVAGGALLTLLLCVGPYNASNVASFLYPNLGGSWRKLGLITGAWSVVLNPLVTGYLGRGPGLKTVCAARTQGGKSQK |
| UniProt | O14842 |
| Protein Data Bank | 5tzy, 5tzr |
| GPCR-HGmod model | O14842 |
| 3D structure model | This structure is from PDB ID 5tzy. |
| BioLiP | BL0380462, BL0380463, BL0380464 |
| Therapeutic Target Database | T25608 |
| ChEMBL | CHEMBL4422 |
| IUPHAR | 225 |
| DrugBank | BE0000688 |
| Name | TAK-875 |
|---|---|
| Molecular formula | C29H32O7S |
| IUPAC name | 2-[(3S)-6-[[3-[2,6-dimethyl-4-(3-methylsulfonylpropoxy)phenyl]phenyl]methoxy]-2,3-dihydro-1-benzofuran-3-yl]acetic acid |
| Molecular weight | 524.628 |
| Hydrogen bond acceptor | 7 |
| Hydrogen bond donor | 1 |
| XlogP | 4.7 |
| Synonyms | AK170559 cc-64 Fasiglifam KS-00001CRE RL00413 [ Show all ] |
| Inchi Key | BZCALJIHZVNMGJ-HSZRJFAPSA-N |
| Inchi ID | InChI=1S/C29H32O7S/c1-19-12-25(34-10-5-11-37(3,32)33)13-20(2)29(19)22-7-4-6-21(14-22)17-35-24-8-9-26-23(15-28(30)31)18-36-27(26)16-24/h4,6-9,12-14,16,23H,5,10-11,15,17-18H2,1-3H3,(H,30,31)/t23-/m1/s1 |
| PubChem CID | 24857286 |
| ChEMBL | CHEMBL1829174 |
| IUPHAR | 6484 |
| BindingDB | 50386790 |
| DrugBank | N/A |
Structure | 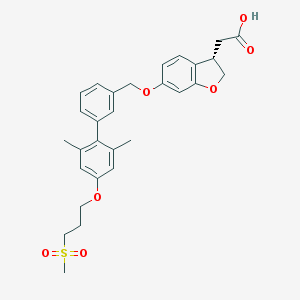 |
| Lipinski's druglikeness | This ligand is heavier than 500 daltons. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Activity | 65.32 % | PMID26482570, PMID26945112, PMID26420383, PMID27020683 | ChEMBL |
| Activity | 76.01 % | PMID26945112, PMID27020683, PMID26420383 | ChEMBL |
| EC50 | 1.9 nM | PMID27994752 | BindingDB,ChEMBL |
| EC50 | 9.3 nM | PMID27074625 | BindingDB |
| EC50 | 9.333 nM | PMID27074625 | ChEMBL |
| EC50 | 14.0 nM | PMID24900210 | BindingDB |
| EC50 | 14.13 nM | PMID21854074 | BindingDB,ChEMBL |
| EC50 | 16.0 nM | PMID22490067 | BindingDB,ChEMBL |
| EC50 | 21.88 nM | PMID22724451 | BindingDB |
| EC50 | 25.7 nM | PMID22724451 | BindingDB |
| EC50 | 26.0 nM | PMID22724451 | BindingDB |
| EC50 | 27.5 nM | PMID27054485 | ChEMBL |
| EC50 | 28.0 nM | PMID27054485 | BindingDB |
| EC50 | 29.6 nM | PMID26482570, PMID27020683, PMID26420383 | ChEMBL |
| EC50 | 30.0 nM | PMID26420383, PMID26482570, PMID27020683 | BindingDB |
| EC50 | 38.6 nM | PMID25481394 | ChEMBL |
| EC50 | 39.0 nM | PMID25481394 | BindingDB |
| EC50 | 42.0 nM | PMID23582779 | BindingDB |
| EC50 | 57.54 nM | PMID27074625 | ChEMBL |
| EC50 | 58.0 nM | PMID27074625 | BindingDB |
| EC50 | 79.4328 nM | PMID21752941, PMID25043059, PMID25787200, PMID24900210 | IUPHAR |
| Emax | 89.0 % | PMID27074625 | ChEMBL |
| Emax | 100.0 % | PMID27054485 | ChEMBL |
| Emax | 108.0 % | PMID27074625 | ChEMBL |
| Ki | 28.84 nM | PMID27074625 | ChEMBL |
| Ki | 29.0 nM | PMID27074625 | BindingDB |
| Ki | 38.0 nM | PMID22490067, PMID24900210 | BindingDB |
| Ratio | 6.89 - | PMID25481394 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218