

You can:
| Name | Urotensin-2 receptor |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | UTS2R |
| Synonym | urotensin II receptor UT receptor UR-II-R UR-2-R UII-R1 [ Show all ] |
| Disease | Asthma Diabetic nephropathy Renal failure |
| Length | 389 |
| Amino acid sequence | MALTPESPSSFPGLAATGSSVPEPPGGPNATLNSSWASPTEPSSLEDLVATGTIGTLLSAMGVVGVVGNAYTLVVTCRSLRAVASMYVYVVNLALADLLYLLSIPFIVATYVTKEWHFGDVGCRVLFGLDFLTMHASIFTLTVMSSERYAAVLRPLDTVQRPKGYRKLLALGTWLLALLLTLPVMLAMRLVRRGPKSLCLPAWGPRAHRAYLTLLFATSIAGPGLLIGLLYARLARAYRRSQRASFKRARRPGARALRLVLGIVLLFWACFLPFWLWQLLAQYHQAPLAPRTARIVNYLTTCLTYGNSCANPFLYTLLTRNYRDHLRGRVRGPGSGGGRGPVPSLQPRARFQRCSGRSLSSCSPQPTDSLVLAPAAPARPAPEGPRAPA |
| UniProt | Q9UKP6 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | Q9UKP6 |
| 3D structure model | This predicted structure model is from GPCR-EXP Q9UKP6. |
| BioLiP | N/A |
| Therapeutic Target Database | T49072 |
| ChEMBL | CHEMBL3764 |
| IUPHAR | 365 |
| DrugBank | N/A |
| Name | CHEMBL573836 |
|---|---|
| Molecular formula | C21H25ClN2O5 |
| IUPAC name | N-[1-(4-chlorophenyl)-4-(dimethylamino)butyl]benzamide;oxalic acid |
| Molecular weight | 420.89 |
| Hydrogen bond acceptor | 6 |
| Hydrogen bond donor | 3 |
| XlogP | None |
| Synonyms | N/A |
| Inchi Key | BNNFDKDTLYXDQK-UHFFFAOYSA-N |
| Inchi ID | InChI=1S/C19H23ClN2O.C2H2O4/c1-22(2)14-6-9-18(15-10-12-17(20)13-11-15)21-19(23)16-7-4-3-5-8-16;3-1(4)2(5)6/h3-5,7-8,10-13,18H,6,9,14H2,1-2H3,(H,21,23);(H,3,4)(H,5,6) |
| PubChem CID | 45481610 |
| ChEMBL | CHEMBL573836 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | N/A |
Structure | 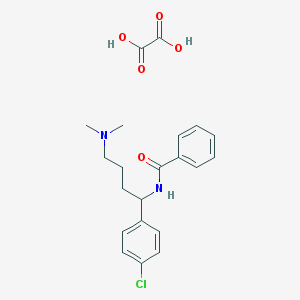 |
| Lipinski's druglikeness | Partition coefficient log P of this ligand is not available. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| EC50 | 3801.89 nM | PMID19481466 | ChEMBL |
| Efficacy | 77.0 % | PMID19481466 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218